Our lab focuses on the study of the Human Immunodeficiency Virus (HIV) the causative agent of Acquired Immunodeficiency Syndrome (AIDS). HIV presents an interesting challenge for two reasons: 1) the virus integrates into the genome of the host cell and can persist for decades, 2) the viral genome is relatively small thus requiring the virus to co-opt various cellular processes to be successful.
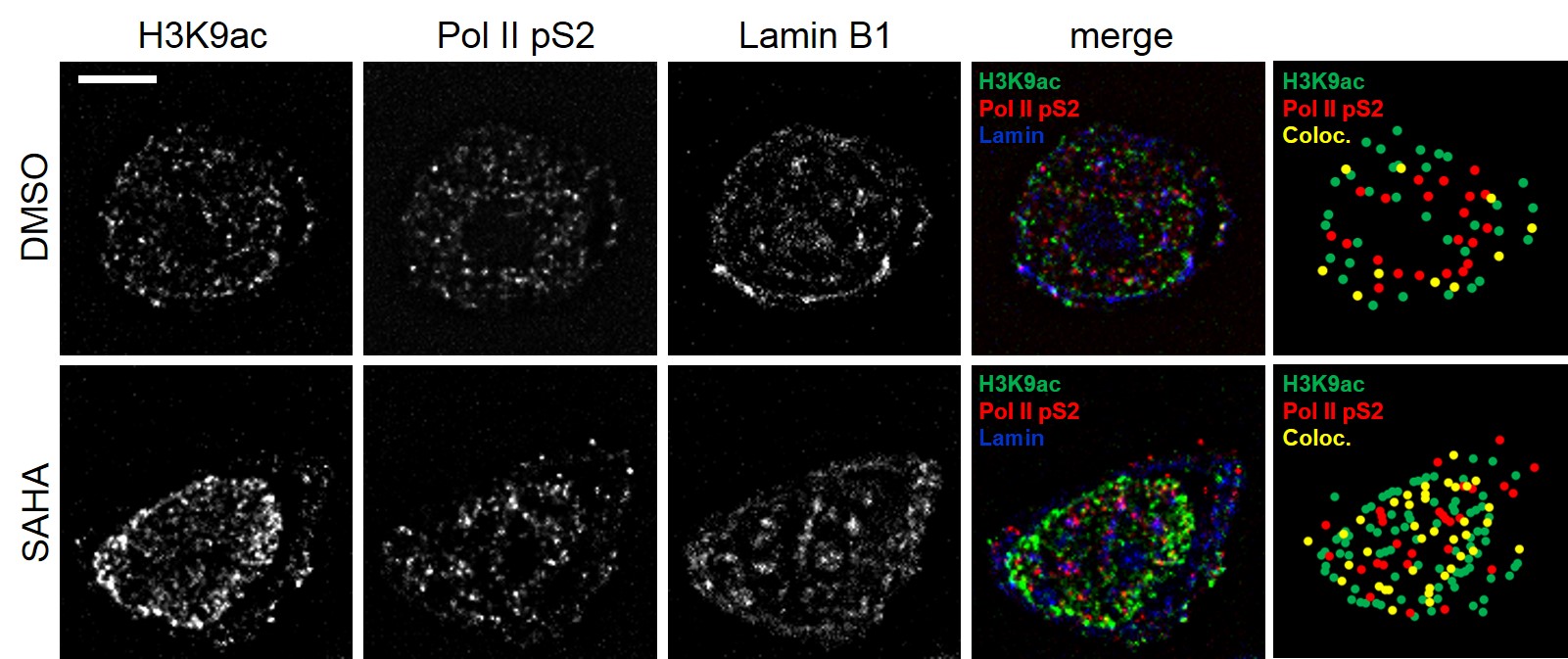
RUNX transcription factors and HIV-1 latency Current therapies are capable of blocking viral replication, preventing new infection and decreasing viral load in the blood to undetectable levels. However therapy does not represent a cure as it does not clear the integrated proviral DNA. HIV infects CD4+ T-cell with a preference for memory T-cells. As memory T-cells are extremely long lived an infected memory cell can potentially carry a silent copy of the virus throughout a patients lifetime and has the potential to restart an active infection after drug therapy ceases. We call this phenomenon latency. A major portion of our time is devoted to studying how the RUNX family of transcription factors contributes to latency and how inhibition of these factors may induce viral transcription and reverse latency.
To study the interaction of transcription factors with DNA a technique called chromatin immunoprecipitation (ChIP) is often used. We have been working to develop new high-resolution microscopy techniques to complement this powerful approach.
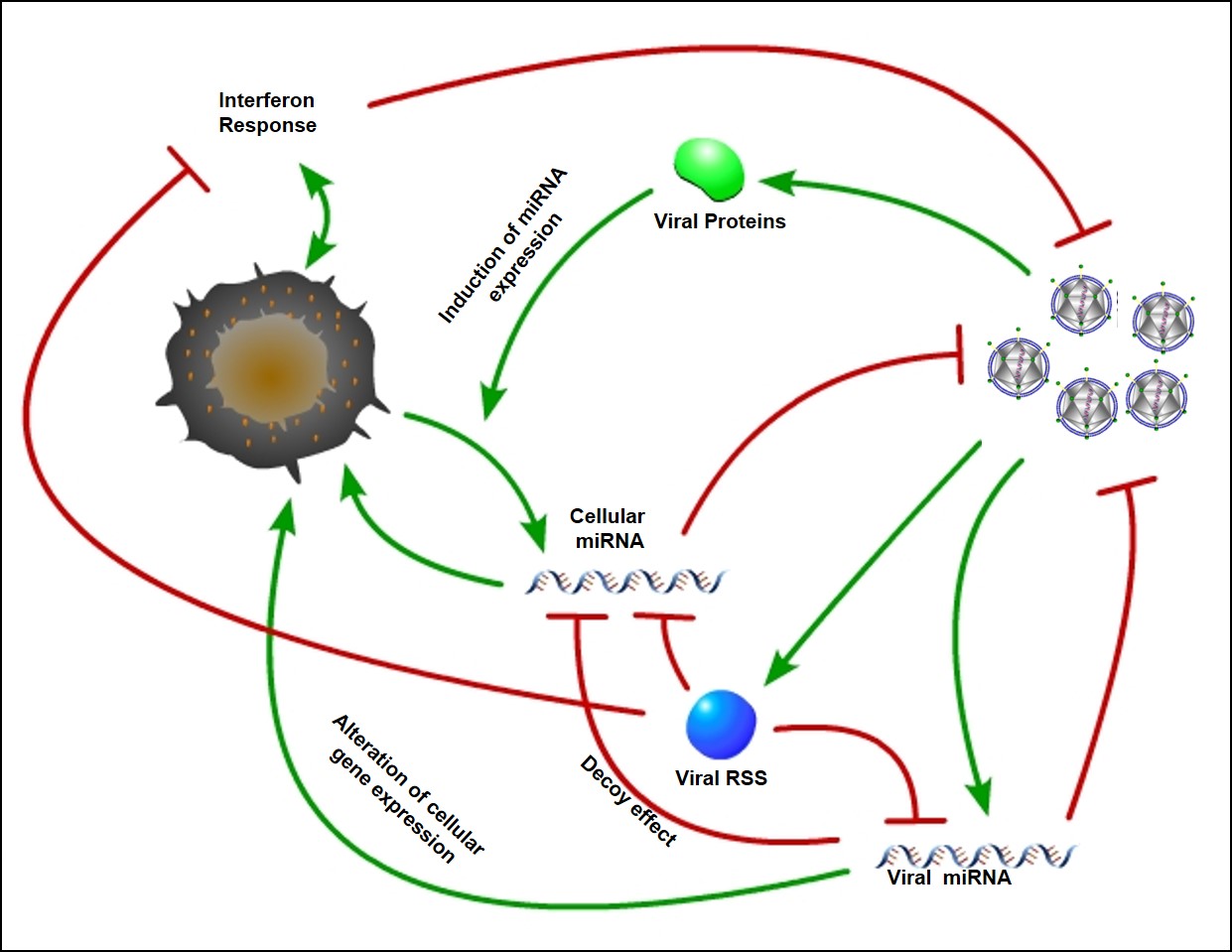
HIV-1 and RNA interference RNA interference (RNAi) can potentially exert anti-viral pressure within a cell. It also represents a very powerful system for controlling gene expression with a cell. As such, it represents a compelling target for a virus to subvert in order to alter host cell gene expression and tune the cellular environment for optimal viral replication. The HIV-1 transcription activator protein Tat has been shown to bind to the RNAi protein Dicer and inhibit the biogenesis of microRNA. We are interested in the specificity of this interaction, how Tat may be altering cellular miRNA expression and what role this plays in the neuropathogenesis of HIV-1
Substance use and HIV-1 There is, unfortunately, a large overlap between HIV-1 infected individuals and with substance use disorders. Our main two interests have brought us naturally into this arena. Many of the miRNA that we see dysregulated by the viral Tat protein seem to have a role to play in neuropathogenesis and we are now studying how these changes are further affected by the presence of morphine. In working with various compounds to address the problem of latency it has become apparent that many drugs of abuse can be a confounding factor. Long term exposure to narcotics or street drugs can even change the way our DNA is packaged in the cell. We are actively working to define how this affects the integrated virus and whether this could be part of (or a barrier to) an eventual cure for HIV.